Ionic vs Covalent
Ionic vs Covalent: What Is the Difference Between Ionic and Covalent Bonds?
Here’s the first thing to know about ionic vs covalent substances. Covalent compounds are held together by covalent bonds. And ionic compounds are help together by ionic bonds. These are two different ways that elements can combine to make substances. So what is the difference between ionic and covalent bonds? You could say that covalent compounds form individual structures of atoms, whereas ionic compounds are based on a repeating pattern of atoms. What? Let’s look at some examples to show the difference between ionic and molecular (another word for covalent) substances.

Take a look at the gas diagram. Each ball represents an individual unit of atoms. We call these molecules. Covalent compounds are better known as molecules. Examples include natural gas (methane) and steam (water vapor). Each ball in the diagram could represent an individual unit of methane, water vapor, or some other molecule. Note there is considerable space between the balls (molecules). Each ball is made of atoms connected by covalent bonds, yet the balls themselves (the molecules) are in no way connected. The tails indicate the gas molecules are flying through space, such as the room you are in. And they bounce off each other when they collide, creating an effect we call pressure.
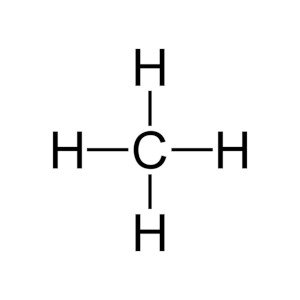
Image that the substance in the gas diagram is methane. Each ball in the gas diagram above represents one methane molecule. Is CH4 ionic or covalent? What is a covalent compound?
Methane, CH4, is a covalent compound with exactly 5 atoms that are linked by covalent bonds. We draw this covalent bonding as a Lewis structure (see diagram). The lines, or sticks, as we say, represent the covalent bonds. There are four bonds from a central carbon (C) linking or bonding it to four hydrogen atoms (H). The methane molecule is this group of 5 atoms connected as such. Methane is known as natural gas. You could image a kitchen stove with a natural gas line. The gas line would deliver an extremely large number of methane molecules, where each molecule is an individual package of the 5 atoms. We could also call molecules covalent compounds, as it means the same thing.
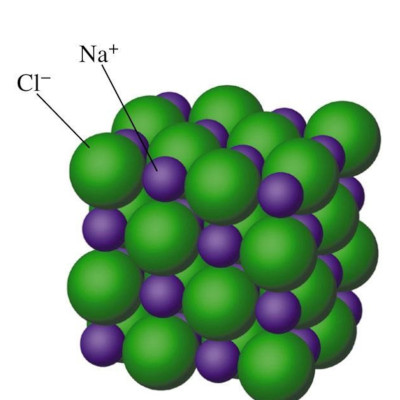
Now take a look at the diagram for an ionic compound, in solid form. First thing, ionic means charged, like plus and minus that attract each other. Note how the atoms alternate green (negatively charged chlorine) and purple (positively charged sodium) in all three directions. It might be obvious that there is one green (negatively charged chlorine) for every purple (positively charged sodium). There is a one-to-one ratio. But is any particular green sphere associated with any particular purple sphere? No, it’s just a pattern that repeats the same in all directions. Ionic compounds, in their usual solid form, always have a repeating pattern of interlocked positive and negative charges like this. We call this ionic compound NaCl (name: sodium chloride, or table salt) because of the one-to-one ratio of the elements Na and Cl.
Unlike covalent compounds like methane above, ionic compounds do NOT:
- have individual units called molecules
- have Lewis structures, which are for molecules only
- have the ability for the atoms to separate, thus becoming a gas like shown for molecules. The plus-minus attractions for ionic compounds necessarily hold the atoms to touch, like a pile of hopelessly interlocked magnets that you could not pull apart with all your strength.
Covalent compounds, or molecules, can be gasses. They can also be liquids and solids. These are called the 3 states of matter. The diagram below shows how the molecules are arranged in solids (left), liquids (middle), and gasses (right). A common example is H2O, called ice when it’s solid, water when it’s liquid, and steam when it’s a gas. Each ball always represents one molecule of H2O, an individual package of 3 atoms. But the molecules themselves are arranged differently in the 3 states of matter. The reason why molecules can be a gas is because they are neutral and don’t have any plus or minus charge. Thus the molecules can separate, unlike the ionic compounds which are locked together by the plus-minus attractions. Molecules can also form solids. It’s like a big city traffic jam, where all the available roadway is filled with cars. Gridlock. And liquid is in between solid and gas, as the gridlock breaks down, the molecules are free to move and flow, but they haven’t yet separated to be a gas.

Ionic compounds are typically found as rocks, minerals, and salts in the Earth. They tend to be solid and dense, due to the strong plus-minus attractions that hold the atoms together. Ionic compounds generally cannot be a gas, as the atoms cannot separate. But can ionic compounds be liquids, with touching yet irregularly arranged atoms? Sure. Under special conditions. Such as lava, which is melted rock, which is ionic compounds. Or molten salt at 1500 degrees Fahrenheit (800 C). These really high melting points for ionic compounds indicate that a lot of energy is required to get them flowing as liquids.
Note that all salts are ionic compounds, but not all ionic compounds are salts.
Covalent vs Ionic: The Simple Answer
So how can you tell if a substance is an ionic compound (rock, salt, mineral) or a covalent compound? That is, how to tell if a substance is ionic vs molecular? The really simple test is:
- Ionic compounds have a metal element (1 or more)
- Covalent compounds (molecules) do not have metal elements
Take a look at the periodic table, color coded to roughly show the metals and other elements. The elements that are not metals are called nonmetals. Very creative, chemistry! There are way more metals than nonmetals on the periodic table. Yet, ionic compounds (which have metals) are relatively simple repeating units. Covalent compounds, or molecules (no metal), can form extremely large and complex structures such as your DNA comprising millions of linked atoms. So there are way more metals than nonmetals, yet there are way fewer ionic compounds compared to covalent compounds (molecules). Living things are made of molecules, as we are far more complex than rocks, at least from a chemistry perspective.
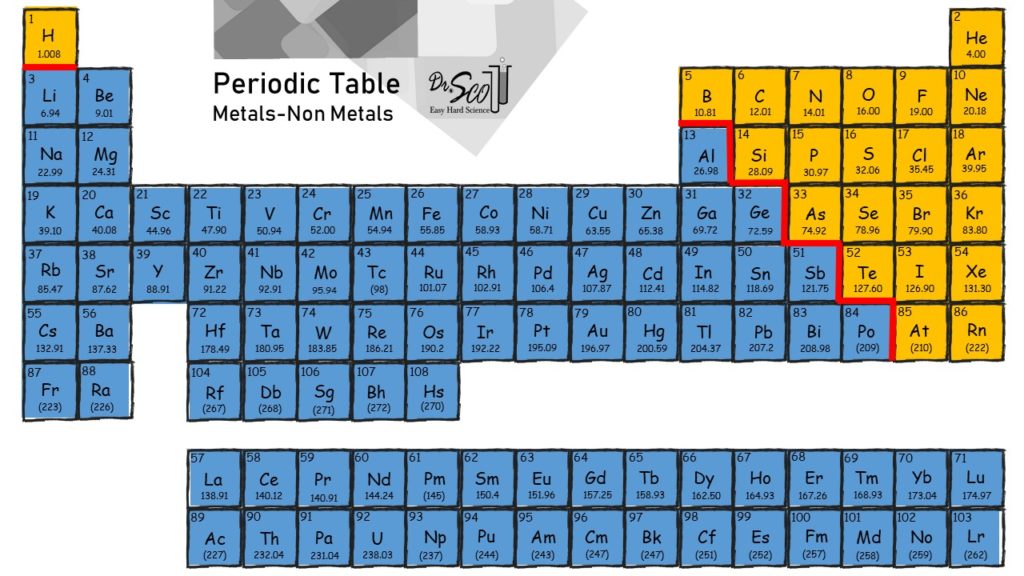
Covalent vs Ionic: The Not-So-Simple Answer
Note I said the periodic table was “roughly” divided into metals and nonmetals. There is not exactly an exact line separating the metals and nonmetals. There is some gray area. For example, an element like silicon (Si, atomic number 14) is a semi-metal (or semiconductor) that can form network covalent bonds. This is kinda like ionic bonding, but also kinda like covalent bonding, but also kinda it’s own thing altogether. Geez. In reality there are more than just two categories: covalent vs ionic. In most normal cases, however, it’s fairly easy to determine if a substance contains a metal, and therefore correctly label it as an ionic compound. Otherwise it’s a molecule (covalent).
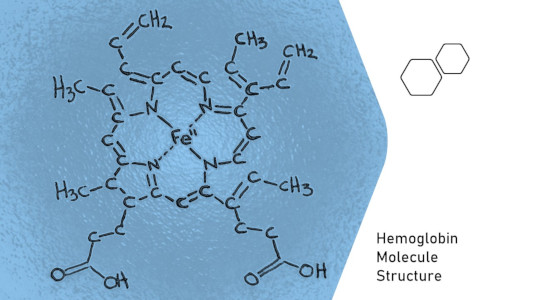
Remember when I said living things are made of molecules? Ever heard that there’s iron in your blood? It’s true. This giant, complex molecule called hemoglobin lives in your blood. It collects oxygen from your lungs and takes it throughout the body. Seems like a good thing. But, the covalent hemoglobin molecule has a single iron atom in it’s center, and this iron atom is what directly attaches to the oxygen. So now molecules can have metals? Sure, but just a little bit of metal and only if the molecule is large and complex. Remember, ionic compounds are more formally defined as simple repeating units whereas molecules are potentially complex, individual structures of atoms. So basically, if it’s complex chemically, then it’s a molecule. That’s not a simple answer, but with some chemistry experience you should start to recognize what is a simple vs more complex substance. That’s why teachers teach about covalent vs ionic in the first place, because it’s a good starting point for learning, even though it’s just a simple categorization into two bins called covalent vs ionic.
This idea of putting metals in molecules won a Nobel Prize in 1912. The French chemist Grignard threw some magnesium metal, which kinda looks like tin foil, into a vat of petrochemicals. Essentially, he used metal to create the molecular version of Lego blocks. He figured out how to combine small molecules to build big, complex molecules resembling those inside of living things. And it required metal to work. We still call this a Gringard reaction today, and it’s the basis of what is called organic chemistry. Organic chemistry in turn is the basis of the modern pharmaceutical industry. You might say this industry is a big and complex thing, much like the medicine molecules they produce, which can be substantially bigger and more complex than hemoglobin.
Covalent vs Ionic: Final Quiz
Look at the diagram for the final quiz. What is it?
Well, it’s art. It’s a drawing, and not a technical drawing like the Lewis structure shown above for methane.
Do you see a simple repeating pattern? No. So it’s probably not an ionic compound.
Does it appear to be a complex, individual structure of atoms? It sure does. I count 19 spheres representing bonded atoms, although there would probably be some more atoms hidden on the backside of the three dimensional structure. It would appear to be an artist’s rendition of a molecule.
