
Finally, the easy way to learn how to find an electron configuration, also known as an orbital diagram or the quantum numbers.
Download and print the black and white pdf. It’s 5 printer-friendly pages. There’s an answer key too in the other pdf file.
There is a simple pattern that you will see in a few minutes by using the below examples. It’s best to learn the topic of electron configurations by example, as it could easily take a million words to describe. Fortunately, we can make a picture of the electron configuration. It’s known as an orbital diagram or formally as an Aufbau diagram.
Hydrogen is element 1 on the periodic table with 1 electron when it’s neutral. The idea is to draw an arrow for each electron, so in this case we just have one arrow to draw. Put the arrow in the lowest box, corresponding to the lowest energy, meaning it is the closest to the nucleus.
We have one upward arrow in the box called 1s. The up arrow means that the electron is spinning half way up, corresponding to a spin quantum number of +1/2. More about the spin quantum number in the next exercise for helium He.
Since there is 1 electron in the box labeled 1s, we say the H electron configuration in orbital notation is 1s1.
The orbital notation can also be interpreted as quantum numbers, where the principal quantum number n is the energy level (1 before the s), the azimuthal quantum number corresponds to the letter s, and the spin quantum number is +1/2. It’s just a much longer way of saying 1s1 as stated above.
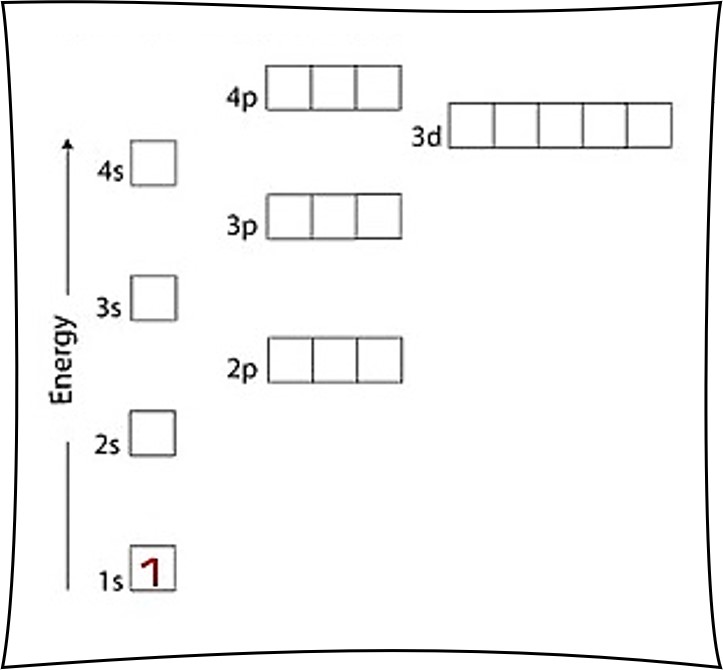
Helium is element 2 with 2 electrons when it’s neutral. The idea is to draw an arrow for each electron, so in this case we have two arrows to draw. There can be up to 2 electrons in any box (representing the electron orbitals). So, we put the two arrows in the first box called the 1s orbital. You could also call it the 1s electron orbital.
The idea is that 2 electrons can be in the same area (an electron orbital) because they spin in opposite directions. Normally, electrons would repel each other because they are negatively charged. But, it’s just a simple law of physics that the opposite happens when charges spin in opposite directions. Hence, 2 electrons can be in the same electron orbital because they spin in opposite directions and attract each other. Generally, it will be the case that electrons live in pairs and spin in opposite directions. One electron spins half way up and has a spin quantum number of +1/2, while the other electron spins half way down and has a spin quantum number of -1/2. Up and down don’t mean anything, nor does the number 1/2. It’s just a funny notation to say that the electrons spin in opposite directions… we just as easily could have said clockwise and counterclockwise instead of +1/2 and -1/2.
The simple logic is that two arrows can go in each box, the first points up and the other points down.
Since there are 2 electrons in the box labeled 1s, we say the He electron configuration in orbital notation is 1s2.
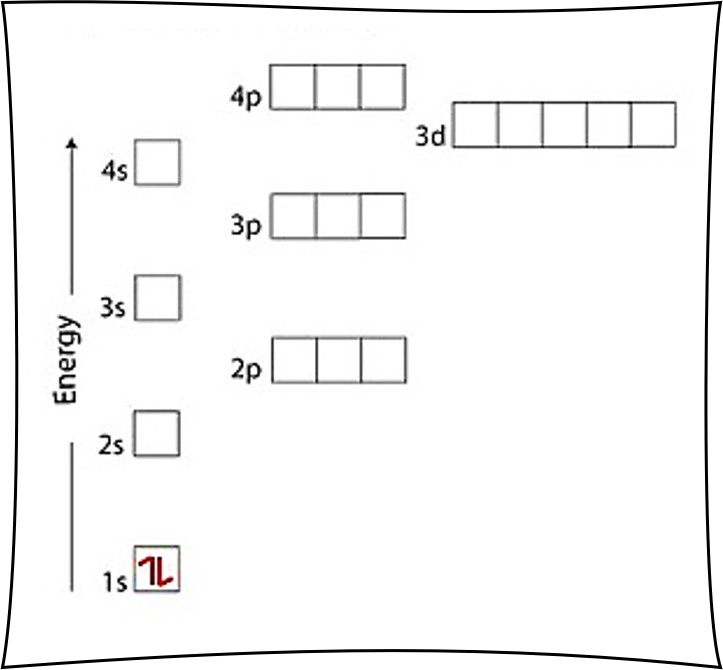
Lithium Li is element 3 with 3 electrons when it’s neutral. After putting 2 arrows in the first box called the 1s orbital, there is still another arrow to draw. It goes in the next box up, called the 2s orbital.
Since there are 2 electrons in the box labeled 1s and also 1 electron in the box labeled 2s, we say the Li electron configuration in orbital notation is 1s22s1.
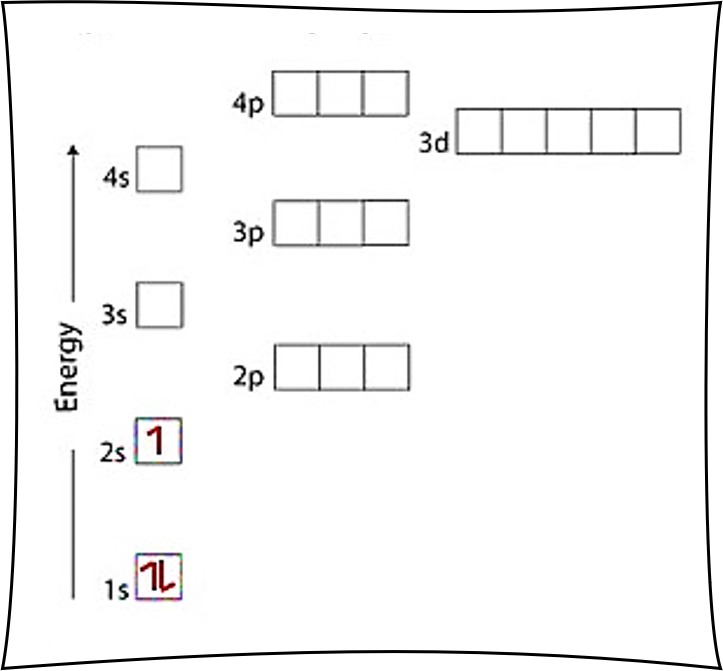
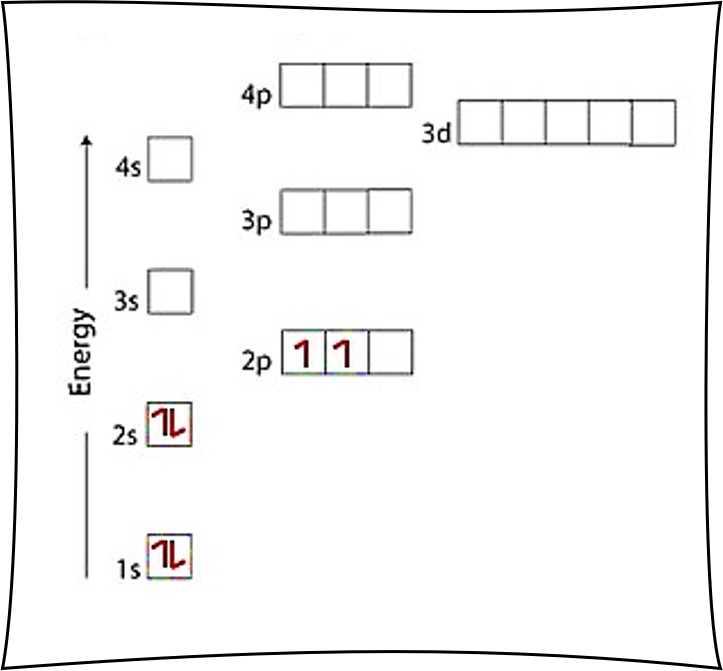
Carbon C is element 6 with 6 electrons when it’s neutral. After putting 2 arrows in the first box called the 1s orbital and another 2 arrows in the second box called the 2s, there are still 2 more electrons to draw.
The arrows always fill from bottom up, so the 2p comes next. Note the 3s is literally above the 2s, but the 2p is the next highest electron orbital to fill. The left-right staggering in the diagram does not matter, we simply go from lowest to highest along the vertical dimension representing distance from the nucleus.
There are 2 arrows to put in the 2p boxes. Note there are 3 boxes, each that can hold 2 electrons. So there is a choice to make. Hund’s Rule states that we put 1 arrow in each box before pairing them up. So, as per Hund’s Rule, there would not be any paired electrons in the 2p. The arrows are in separate boxes of the 2p, and both point up.
Since there are 2 electrons in the box labeled 1s, 2 electrons in the box labeled 2s, and also 2 electrons in the boxes labeled 2p we say the C electron configuration in orbital notation is 1s22s22p2.
Oxygen O is element 8 with 8 electrons when it’s neutral. The 1s orbital is full, the 2s orbital is full, and there are 4 electrons to draw in the 3 boxes in the 2p orbital. As per Hund’s Rule, there would be 3 arrows pointing up in the 2p orbital and 1 pointing down. So there is a pair of electrons in the first box of the 2p only.
Since there are 2 electrons in the box labeled 1s, 2 electrons in the box labeled 2s, and also 4 electrons in the boxes labeled 2p we say the O electron configuration in orbital notation is 1s22s22p4.
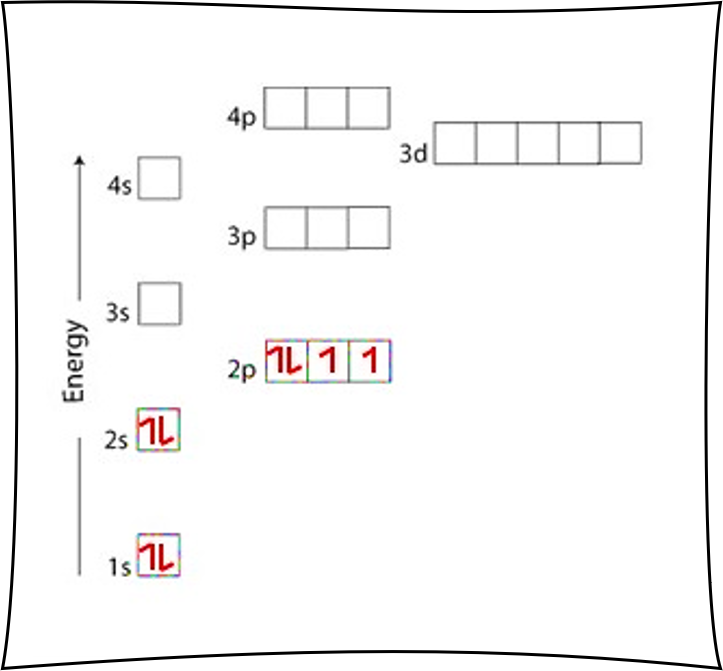
Argon Ar is element 18 with 18 electrons when it’s neutral. The 1s orbital is full, the 2s orbital is full, the 2p orbital is full, the 3s orbital is full, and the 3p orbital is full. Fill the orbitals in this order 1s then 2s then 2p then 3s then 3p, from bottom to top on the orbital diagram.
Note this is a special case that Hund’s Rule doesn’t really matter, since the 2p and 3p boxes are completely full of arrows. It doesn’t matter what order they are drawn, since they are just full. Still, it’s a good idea to just always respect Hund’s Rule regardless and let whatever is going to happen happen.
Since there are 2 electrons in the box labeled 1s, 2 electrons in the box labeled 2s, 6 electrons in the boxes labeled 2p, 2 electrons in the box labeled 3s, and 6 electrons in the boxes labeled 3p, we say the Ar electron configuration in orbital notation is 1s22s22p63s23p6.
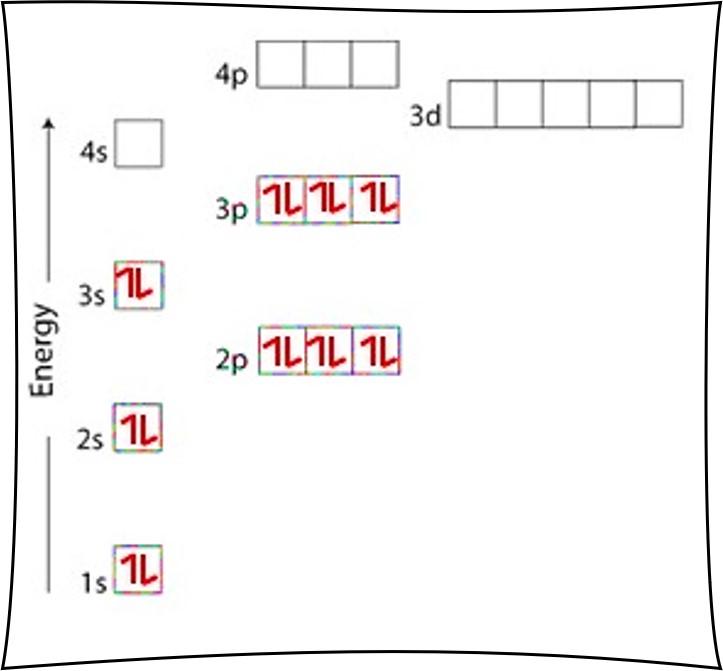
Iron Fe is element 26 with 26 electrons when it’s neutral. The first 18 electrons fill as for Argon Ar in the previous example. Next comes the 4s orbital, which is full with 2 electrons.
We now place the remaining 6 electrons in the 3d orbital, as per Hund’s Rule. Draw 5 arrows up, one in each of the 5 boxes. The 6th arrow is drawn down in the first box.
Since there are 2 electrons in the box labeled 1s, 2 electrons in the box labeled 2s, 6 electrons in the boxes labeled 2p, 2 electrons in the box labeled 3s, 6 electrons in the boxes labeled 3p, 2 electrons in the box labeled 4s, and 6 electrons in the boxes labeled 3d we say the Fe electron configuration in orbital notation is 1s22s22p63s23p64s23d6.
Now, there is some funny business here in that the 4s orbital in the fourth energy level fills before the 3d orbital in the third energy level. The d (and f orbitals too) are a bit out of order. It’s okay to group the numbers, writing the orbital notation as 1s22s22p63s23p63d64s2 instead of 1s22s22p63s23p64s23d6. Both are correct.
Another issue is that the orbital notation is getting long. There is a shortcut. Note the argon electron configuration above is 1s22s22p63s23p6 which could simply be called [Ar]. We can bracket any noble gas as a shortcut. We could also say the Fe electron configuration in orbital notation is [Ar]4s23d6 or even [Ar]3d64s2.
![ground state electron configuration for iron 1s22s22p63s23p64s23d6 [Ar]4s23d6](https://learnwithdrscott.com/wp-content/uploads/2020/11/ground-state-electron-configuration-for-iron-Ver2.jpg)
Learn all the chemistry topics in a free chemistry course ($299 value) now!