This page has a printable electronegativity table, also called an electronegativity chart.
The electronegativity definition and electronegativity trend are described below.
Prefer YouTube style video lessons? Get unlimited access for a low monthly membership fee.
Download and print the black and white pdf. It’s 1 printer-friendly page.
Electronegativity is the ability for an atom to gain an electron.
Yawn!
We are talking about electron piracy here. Electronegative elements are like pirates. They steal, plunder, and take electrons from less electronegative elements.
Generally, more electronegative atoms take electrons from less electronegative atoms. It’s all relative.

Electronegativity is a concept developed by Linus Pauling, a rather famous chemist who won the Nobel Prize twice (once for chemistry, once for peace).
The idea is to distill all the knowledge of chemistry into a single number, a scale, that ranges from 0 to 4. Each element has an electronegativity value from 0 to 4. We usually show electronegativity on the periodic table, as shown in the image below (same as the printable download above).
Electronegativity is useful for determining the polarity in chemical bonds, resulting in the concept that “Like Attracts Like” which is commonly taught in biology and chemistry.

Can you find the highest value of 4.0? Look at F, fluorine, in the upper right of the periodic table.
Can you find the lowest value? There is no zero shown on the electronegativity chart. In the bottom left corner, elements Fr (francium) and Cs (cesium) have values of 0.7.
The electronegativity trend goes from the lower left of the periodic table to the upper right. Excluding the noble gasses (more on that below).
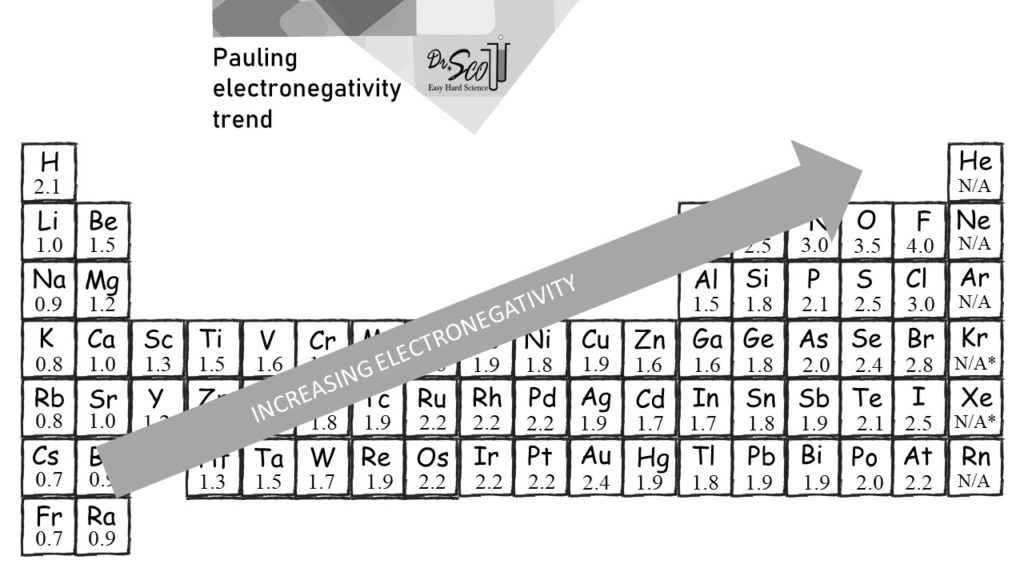
The electronegativity trend is generally more important than the actual numbers. It’s all relative, and generally you only need to know which of two elements is more electronegative. The numbers themselves are rarely used. It’s mostly just a scale to order the elements.
For example, which element is more electronegative: O oxygen or S sulfur? O is above S on the periodic table, and the trend increases to the upper right. So you can be relatively certain that O is more electronegative than S. The numbers on the above chart confirm this.
Noble gasses don’t generally have electronegativity. You should probably say that a noble gas has zero electronegativity, because they don’t form bonds and therefore must not want electrons. But, the calculation of electronegativity from experimental data is a bit complicated.
Values for the noble gasses He, Ne, Ar, and Rn are listed as N/A (not applicable). These noble gasses won’t bond. Therefore an experiment to measure their bond energies is not possible, and there is no way to actually calculate the electronegativity. It should be zero, but there are no data. As such, the best answer is N/A.
It gets worse. Noble gases Kr and Xe can be made to react in a modern lab. There are extremely few such reactions. Based on this very limited experiment data, of just a few reactions, Kr and Xe can have electronegativity values calculated. They are actually rather high (3.0 for Kr and 2.6 for Xe). These numbers are just special cases, and you certainly should not associate any meaning to them. As such, the table shown here lists Kr and Xe as N/A*. The asterisk is how scientists say that we happen to have an experimentally accurate number, yet the experiment itself is pure garbage.
Most electronegativity charts just don’t show the noble gasses. Because it’s complicated and moreover irrelevant. What is stated here is true. Yet if anybody ever asks you about the electronegativity of noble gasses, the most appropriate response would be to give them a funny look. It just doesn’t make any sense in the larger context of chemistry.
The electronegativity trend is not so clear for the transition metals. These are the elements in the middle of the periodic table, set down lower than on the sides. Notably, gold (Au), a mostly inert metal, has a higher electronegativity than all other metal elements.
Transition metals are usually described as having a “sea of loosely held valence electrons.” This explains their electrical conductivity, for example. So, the concept of electronegativity, the stealing of electrons, isn’t so relevant for transition metals.
Electronegativity works best for the s-block (two leftmost columns) and the nonmetals (upper right of the periodic table, excluding the noble gasses). It explains the formation of ionic bonds when these elements combine.