The CO2 Lewis structure has two double bonds going from carbon to the oxygen atoms. According to the octet rule, each oxygen atom needs to bond twice and the carbon atom needs to bond four times.
Let’s think in terms of dots to make the CO2 Lewis structure.
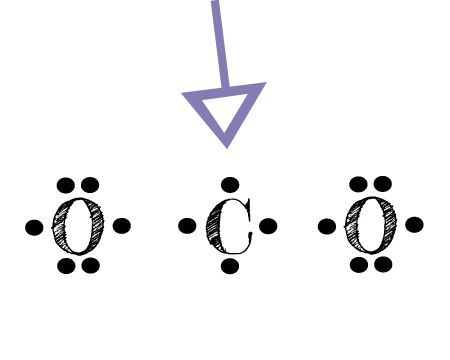
Carbon has four valence electrons that form a total of four bonds. So carbon is shown with four dots around it. Oxygen needs just two bonds, represented as the lone dots to the left and right of the O atoms. The pairs of dots above and below the O’s won’t bond.
The first thing about the CO2 Lewis structure is to put carbon in the center. Make both O atoms connect to C. There won’t be any bonds between the O’s directly. As a rule, carbon is always going to be in the center, and the other atoms connect to it.
Second, connect the lone dots on each O to the C in the center. Each O needs to bond twice. And carbon needs four bonds. So it works out that C bonds with each O twice.
The bonds appear as the parallel lines between the C and O atoms. These are called double bonds.
Each O is surrounded by four dots and two sticks or lines, representing another 4 electrons in its double bond. So each O is surrounded by 8 total valence electrons, giving it an octet and making it stable. Carbon has four bonds, in this case present as two double bonds. So carbon also has 8 valence electrons.
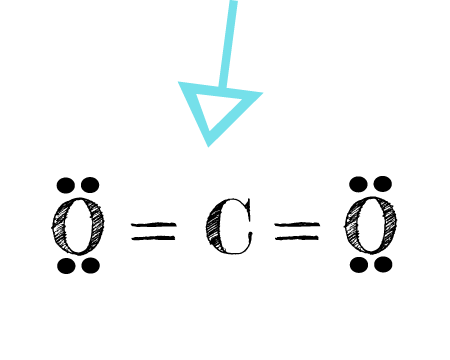
The CO2 Lewis structure is symmetric. Generally, small symmetric molecules are nonpolar.
CO2 is a nonpolar substance, meaning it tends to be a gas. CO2 has a rather low boiling point of around -80 ℃ or -100 ℉. It can be liquified and even frozen solid with special machinery to produce “dry ice.” Dry ice exhibits a temperature around the boiling point mentioned previously. It’s used in the chem lab when regular ice doesn’t get things cold enough.
Click for more advanced knowledge of CO2 properties, including additional information about its Lewis structure.
Also, CO2 is that chemical that humans breathe out and trees breathe in. Importantly, nonpolar substances like CO2 are gasses at normal atmospheric temperatures. Lesser known, atmospheric CO2 also absorbs into oceans, where it can form carbonic acid, which can interfere with animals that produce calcium carbonate shells.