
Molar mass means just that. It’s the mass, or weight, of a mole. For the record, a mole is generally about a palmful of a substance. The size and weight of a mole is different for every substance.
Note that mass and weight are the same thing on planet Earth. So it’s okay to use the words mass and weight interchangeably to describe a mole. Common terms include molar mass, atomic mass, atomic weight, molecular weight (abbreviated MW), and formula weight. It’s trendy to say “molar mass” because the word “weight” is sometimes viewed as a bad word by many scientists, and not all substances are molecules or individual atoms.
Download and print the black and white Molar Mass Worksheet pdf. There’s an answer key too in the other pdf file.
There are examples and explanations below.
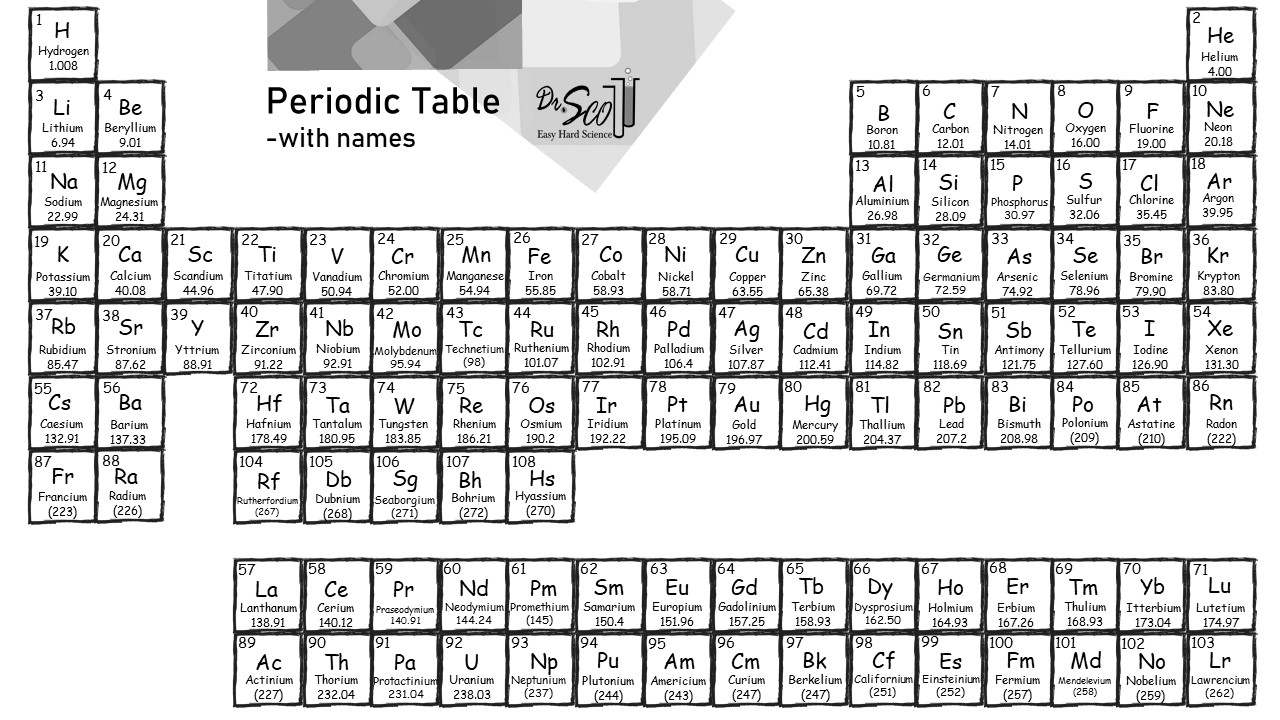
The molar mass of sodium is found on the periodic table. For element Na (sodium), find the number with the decimal point. It’s 22.99 not 11. So, a mole should weight 22.99 grams. Add the unit grams at the end of the number with the decimal point. This is the molar mass, 22.99 grams.


The problem is that 22.99 grams is not a very nice number. It’s practically 23 grams. Moreover, some scale, such as the one shown, are only accurate to the nearest gram. This scale doesn’t understand the concept of 22.99 grams. It only “sees” 22 grams or 23 grams, and nothing in between. So, as a practical matter, it’s much better to round the number to 23 grams. The molar mass of sodium is 23 grams.
Can we really get away with this rounding? In short, yes, Note the official molar mass of sodium is actually 22.98976928. This still rounds to about 23 grams. Whenever a decimal appears in science, it means the number is an average that has been rounded. It’s okay to round further in most cases.
Also, note that the atomic mass of sodium is the weight of one sodium atom. It would be approximately 23 a.m.u. (atomic mass units). The weight of one atom and the weight of a mole are the same number, just different units.
To get the molar mass of NaCl, add Na + Cl using the numbers on the periodic table. Na rounds to 23 grams. Chlorine doesn’t round so cleanly. It’s half way between 35 and 36. Perhaps we should keep one decimal point and call it 35.5.
Add 23 grams for Na plus 35.5 grams for Cl to get the molar mass of NaCl, which is 58.5 grams.
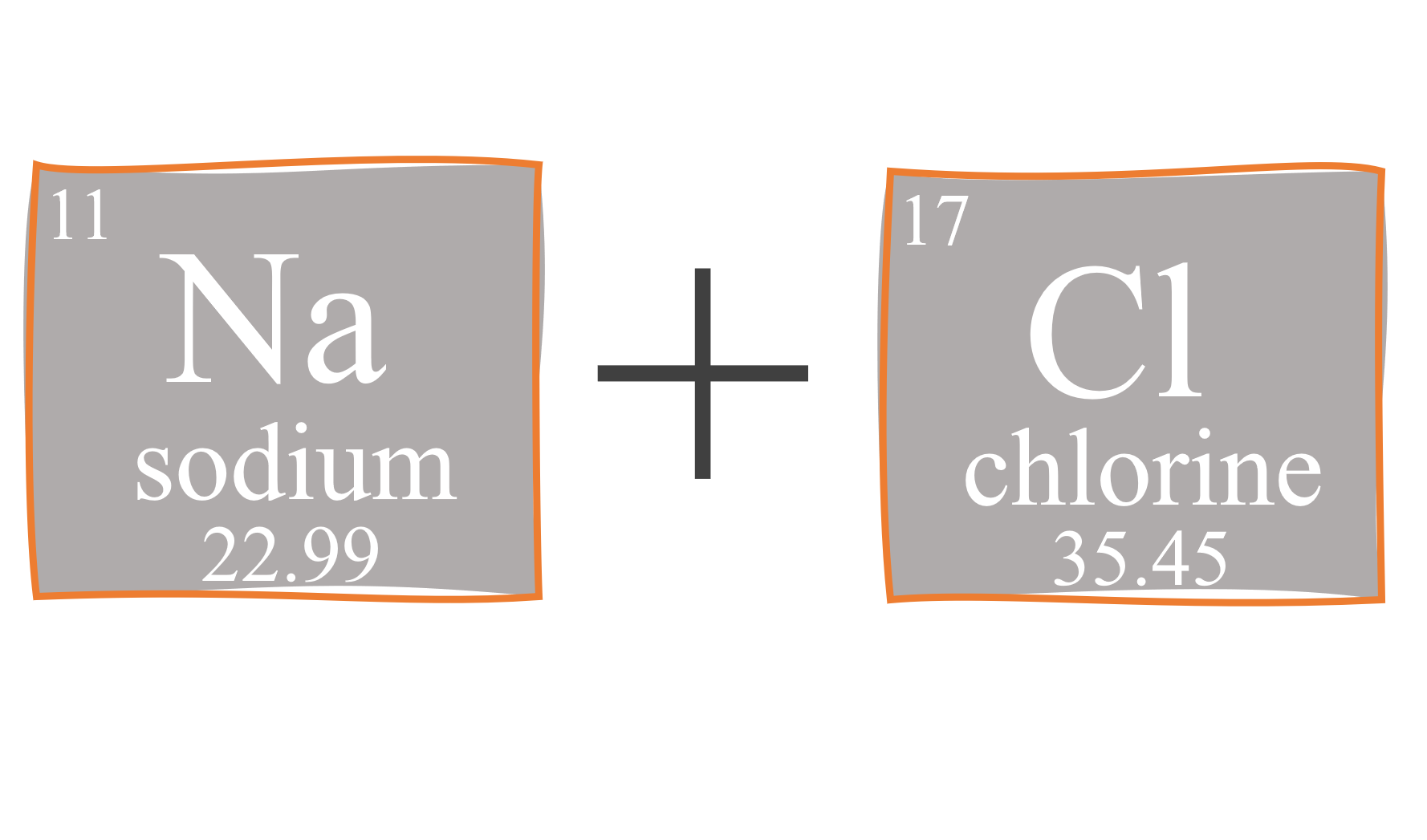
Oxygen, the gas in the atmosphere, is actually a pair of oxygen atoms. The word oxygen has two meanings. Oxygen is letter O on the periodic table, but oxygen is also a gas you breathe with the formula O2.
The periodic table is literal. It says that one O atom weighs 16 grams. Oxygen the gas is O2, which has 2 O atoms weighing 16 grams each. In short, double 16 to get the molar mass of O2 as 32 grams.
This strange situation happens for exactly 7 elements. Hydrogen is H2, nitrogen is N2 , fluorine is F2, chlorine is Cl2, bromine is Br2, iodine is I2, and oxygen is O2. They are called diatomic elements. The molar mass is twice that shown on the periodic table, because these 7 elements exist as pairs of atoms. Again, the mass of O2 is 32 grams, double the 16 grams on the periodic table.
To get the molar mass of Fe2O3, first find Fe (55.9 grams) and O (16 grams) on the periodic table.
Double the iron mass (multiply by 2, the subscript on Fe in Fe2O3) to get 55.9 grams x 2 = 111.8 grams. Triple the oxygen mass (multiply by 3, the subscript on O in Fe2O3) to get 16 grams x 3 = 48 grams. The Fe2O3 molar mass is the sum 111.8 + 48 = 159.8 grams.
Note you better get a calculator to work out the molar mass of Fe2O3.
Many chemicals, especially hydrocarbons with just elements C and H, have common names. You have to know, or look it up, that butane has the formula C4H10 to get started.
The C4H10 molar mass is found my taking 4 x 12 grams (for carbon) and 10 x 1 gram (for hydrogen). The C4H10 molar mass is 48 + 10 = 58 grams.
You would have to know that ethanol is C2H5OH to get started. Ethanol is an alcohol, and chemists always write OH at the end of an alcohol. For this problem, you should think in terms of C2H6O, even though it’s never written that way.
There are 2 carbons (12 grams each, 24 grams total), 6 hydrogens (1 gram each, 6 grams total), and one atom of oxygen (16 grams). The molar mass of ethanol is 24 + 6 + 16 = 46 grams.
The molar mass of CaCO3 is 40 (for Ca) plus 12 (for C) plus thrice 16 (for 3 O atoms). That’s 40 + 12 + 48 = 100 grams for the molar mass of CaCO3 ,calcium carbonate.
Calcium carbonate, the major component of limestone, can form spectacular formations such as a petrified waterfall.
Add the masses for Na, Cl, and three times O. That’s 23 + 35.5 + 48 = 106.5 grams.
The molar mass of KClO3 is 39 grams (for K) plus 35.5 grams (for Cl) plus 16 x 3 = 48 grams (for O). The molar mass of KClO3 is 39 + 35.5 + 48 = 122.5 grams.
Note for the KClO3 molar mass of and also NaClO3 above, we rounded chlorine to 35.5 grams (not 36 grams) to avoid about half a gram of rounding error.
This is very similar to Example 1, the molar mass of sodium (Na, the neutral atom). In fact, the answer is the same.
A sodium ion is denoted Na+. The word ion means it has charge, written to the upper right as being positive in this case. Sodium ion has lost an electron relative to neutral sodium. Electrons, however, don’t have weight or mass. So the charge doesn’t change the molar mass.
The molar mass of Na+ is the same as Na, which was 23 grams in Example 1.